ABPI Resources - interactive resources and animations for an extensive list of biological topics.
BBC Wonder - dedicated BBC section to science and answering fundamental questions in all science areas.
Bioethics Education Project - activities and discussions for a wide range of bio ethical issues.
Bio Digital - a 3D interactive human body and anatomy experience.
Biology 4 All - bank of revision resources and quizzes for a variety of A level and wider reading topics.
Biology Guide - notes on all topics of biology in an easy bullet point form.
Biology Practicals - notes and diagrams for practicals covering the A Level specification.
e-Bug - information and games on microbes and antibiotic resistance.
Ed's Basic Histology Gallery - a collection of annotated histology images from under the microscope.
Gray's Anatomy - an online version of the book showing the anatomy of the whole body.
Human Physiology - lecture notes on human physiology and anatomy.
Inner Body - interactive model of human body systems.
Interactive Biology - contains extensive amounts of videos discussing A level biology topics.
Lens Shopper - interactive human anatomy of the eye.
Lone Star College Animations - biological animations.
Molecular Visualisation - 3D interactive biological molecules.
Spolem - online biology lessons and free worksheets.
The A Level Biologist - brilliant notes, discussions and videos regarding A Level biology.
What does it mean to be human? - an interactive human evolution timeline.
Your Genes, Your Health - a multimedia guide to genetic disorders.
Monday, 5 October 2015
Biological Importance of pH
pH - What's all the fuss about?
pH (potential of hydrogen) is a measure of the degree of acidity or alkalinity of a solution - representing the concentration on H+ ions on a log scale.
- The availability of mineral ions for uptake by plants is affected by pH of the soil. Therefore, the productivity of crops and yield is also dependent on pH.
- pH of stomach acid is acidic to kill and remove any bacteria - a primary defence mechanism.
- pH affects the tertiary shapes of enzymes as the hydrogen bonds holding it together breaks. Thus, the active site shape changes and in no longer complementary to the substrate and no enzyme structure complexes are formed; denaturing the enzymes.
- CO2, a product of respiration dissolves in the blood plasma to become carbonic acid - a weak acid lowering the blood's pH.
Biological Buffers in the Blood
Acidaemia - when the pH of the blood falls below normal.
Alkalaemia - when pH of the blood rises above normal.
The buffer in blood consists of two main parts:
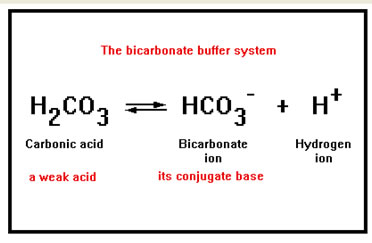
- HYDROGEN CARBONATE IONS - relies on the following equilibrium being to the left. Thus, an increase in H+ ions means equilibrium shifts to the left, producing more carbonic acid. This then dissociates further to water and CO2; reducing acidity.
- BLOOD PROTEINS (haemoglobin and plasma proteins) - proteins are made up of amino acids with a structure like this:
Role of the Lungs: CO2 dissolves in the blood to become a weak carbonic acid. Thus, it is important for CO2 to be removed by the lungs after respiration to prevent the blood from becoming too acidic.
Role of the Kidneys:Acidity of food intake also has an effect on the pH of blood. The kidneys removes acid via excretion in the distal covulated tube of the nephrons of the kidney.
- Any carbonic acid produced in the tubule is rapidly dissociated to H+ and HCO3- by the enzyme carbonic anhydrase.
- The HCO3 combines with Na+ from step 4 to produce NaHCO3 and sodium is released back into the blood.
- H+ ions are actively secreted into the tubule.
- H+ react with Na2HPO4 producing NaH2PO4+ and Na+.
- The Na+ is actively reabsorbed back into the blood.
* Hydrogen ions are buffered into the urine.
* Sodium is not lost to the body.
Thompson H, Manuel J. 1996, Further Studies for Health. London, Hodder Headline, ISBN: 9780340620557
Subscribe to:
Comments (Atom)


